2024 CMT Road Map to Clinical Trials
*scroll down to view all videos
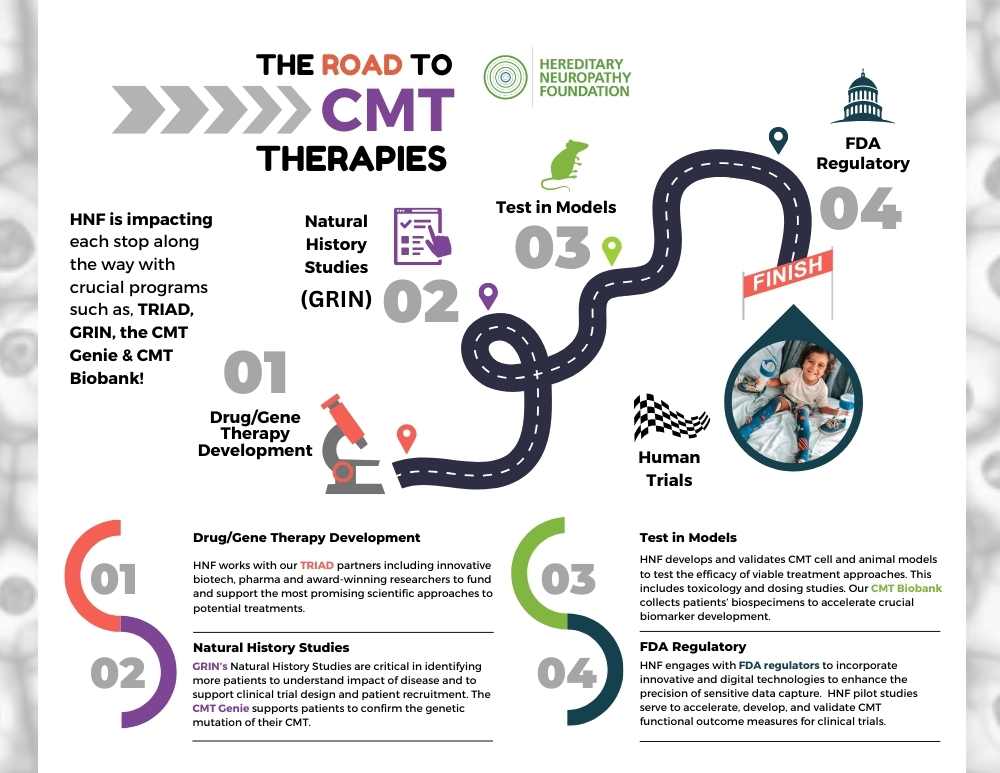
As we look ahead to emerging CMT clinical trials, the road map has never been clearer. At the Hereditary Neuropathy Foundation, we understand the importance of approaching each critical step with the utmost efficiency, competency and urgency. For any medical treatment to reach human clinical trials, there is a precise formula and path we all must follow. This is the 4 step pathway HNF’s vital initiatives are designed around. And this is why HNF’s TRIAD, GRIN, CMT Genie, and CMT Biobank are pivotal to each and every step.
But what exactly are the 4 steps on the road to human trials?
Step 1 is Drug/Gene Therapy Development
HNF works with our TRIAD partners, including innovative biotech, pharma, and award-winning researchers, to fund and support the most promising scientific approaches to potential treatments. In 2023, we conducted Repurposed Drug Screenings for 10 subtypes of CMT, and accomplished 9 potential drugs for three CMT subtypes. We continue to advance our gene therapy program for 2 subtypes of CMT.
Step 2 is Natural History Studies
These studies are a critical requirement for any human clinical trial. GRIN’s Natural History Studies are vital to characterizing the different manifestations of CMT and understanding the progression of the disease and loss of function over time. They allow us to identify patients for recruitment and design successful clinical trials.
Continue
In 2023, HNF has grown GRIN to over 5000+ participants with 41 CMT mutations in 60 countries, and we’ve launched a pediatric natural history study. Our latest product is the CMT Pain Management Guide.
HNF’s CMT Genie supports patients to confirm the genetic mutation of their CMT so that their symptoms and lived experiences can be correlated to their specific mutation. Since most therapeutic approaches target specific CMT subtypes, genetic testing is more important than ever.
In 2023, over 300 patients participated in the CMT Genie, with over 130 individuals testing positive for some subtype of CMT. For those whose results were negative or inconclusive, HNF continues to support our community with the next steps and additional options that will help lead to the answers we all deserve.
Step 3 is Testing in Models
HNF develops and validates CMT cell and animal models to test the efficacy of viable treatment approaches. This includes toxicology and dosing studies. In 2023, we’ve established 2 Rat Models, and 3 Cell Models. Our new CMT Biobank collects patients’ biospecimens to accelerate crucial biomarker development.
Step 4: FDA Regulatory
HNF continues to engage with FDA regulators to incorporate innovative and digital technologies to enhance the precision of sensitive data capture. Our pilot studies serve to accelerate, develop, and validate both traditional and innovative methods for CMT functional outcome measures leading to clinical trial approval.
Continue
We continue to share the groundbreaking CMT Report from our 2018 Patient-focused Drug Development Meeting.
Through sharing these findings with the FDA, biotech, and Pharma, we ensure that the patients’ experiences, perspectives, and needs can be used to develop new treatments and support their approval by the (FDA).
HNF will continue down the road to human clinical trials as patients, advocates, researchers, and innovators. Just like CMT has its challenges, so does our journey to treatments. We thank you from the bottom of our hearts for your support in 2023 and continued dedication in 2024 and beyond.
We need your support and donations to continue to fuel our efforts to reach our ultimate finish line: successful human clinical trials for CMT.
The Hereditary Neuropathy Foundation invites you to explore our CMT Roadmap and discover how, together, we’re making an impact that matters.
Our Mission
Our mission is to increase awareness and accurate diagnosis of Charcot-Marie-Tooth (CMT) and related inherited neuropathies, support people living with CMT and their families with critical information to improve quality of life, and fund research that will lead to treatments and cures
How we advance Clinical Trials
HNF continues to be instrumental in advancing clinical trials by:
- Providing valuable patient insight
- Developing innovative data collection
- Consulting industry-sponsored clinical trial design
- Informing the FDA for decision-making and oversight during drug development and commercialization of drugs and gene therapies for CMT.
- Advocating for regulatory initiatives
How has HNF advanced & supported clinical trials for CMT?
HNF has developed a collaborative team of experienced CMT experts, patient advocates and clinical trial resources to support clinical trials for all types of CMT.
HNF’s Global Registry for Inherited Neuropathies (GRIN) helps to promote the needs and priorities of patients through IRB-approved studies.
These studies have and continue to generate robust data on genotype (CMT subtypes) correlated to phenotype (signs & symptoms) from our Natural History Study as well as generated data from ClinGen surveys that are recognized as valid sources of patient data by FDA.
What are our Industry Partners saying?
“Initiatives such as the HNF registry bring together critical data from patients, caregivers and clinicians, including genetic tests and electronic health records. This information can help inform clinical trials of therapies for the 150,000 people in the US who are living with Charcot-Marie-Tooth disease and have no treatment options available,”
“GRIN data has been essential in clinical trial protocol design, site selection, and efficient patient recruitment for the pivotal Phase III clinical study of our lead asset PXT3003, the PREMIER trial, in development for the most common type of CMT, CMT1A.
Latest on HNF Industry Partnerships Clinical Trials
SORD Deficiency
A clinical-stage biopharmaceutical company that has developed a novel molecule to treat the newly discovered type 2 CMT gene which causes SORD deficiency. Experts believe that SORD deficiency could very well become the most common subtype of CMT 2. HNF has been instrumental in helping to facilitate and accelerate the approval of their clinical lead product AT-007, a Aldose Reductase Inhibitor (ARI) currently in clinical trials.
“Initiatives such as the HNF registry bring together critical data from patients, caregivers and clinicians, including genetic tests and electronic health records. This information can help inform clinical trials of therapies for the 150,000 people in the US who are living with Charcot-Marie-Tooth disease and have no treatment options available,” said Shoshana Shendelman, CEO & Founder of Applied Therapeutics.
Pharnext Pharmaceuticals
A late stage clinical biopharmaceutical company has developed a novel synergistic drug combination to treat the most common type of CMT, CMT1A. The drug Syngility is a fixed-dose in combination of baclofen, naltrexone and sorbitol to downregulate the overexpression of PMP22 protein, leading to improvement of neuronal signaling in dysfunctional peripheral nerves which is the primary pathophysiology problem for CMT1A. HNF has been in partnership with Pharnext since 2012 and is a key contributor to supporting the development and commercialization of Syngility. Expected results of their pivotal phase III is expected by Q4, 2023.
“GRIN data has been essential in clinical trial protocol design, site selection, and efficient patient recruitment for the pivotal Phase III clinical study of our lead asset PXT3003, the PREMIER trial, in development for the most common type of CMT, CMT1A. In addition, the robust data collected in GRIN, together with the ongoing CMT&Me digital survey, will enable a better understanding of the burden of disease, comorbidities, and critical patient demographics as we move forward to a potential commercialization of PXT3003 if approved by the FDA. Topline data of the PREMIER trial are expected in the last quarter of 2023.” Xavier Paoli M.Sc. Pharnext, Chief Operating Officer
We believe this clinical trial has the potential to be a crucial turning point in the effort to finally provide an efficacious treatment for patients suffering from CMT1A,” said Daniel Cohen, M.D., Ph.D., co-founder and CEO of Pharnext. “Today’s therapeutic options are very limited and mostly palliative in nature. Our PLEODRUG® PXT3003 has already demonstrated safety, tolerability and improvements in CMT1A patient disability in a Phase 2 trial. Given this positive safety assessment by the DSMB, we are hopeful to bring this much needed potential therapy to patients suffering from this debilitating condition upon completion of this Phase 3 trial.”
Join GRIN!
Without your participation, researchers won’t have the essential and necessary patient information to develop drugs, gene therapies, and clinical trials for Charcot-Marie-Tooth and other Inherited Neuropathies!
Genetic Testing – CMT Genie
Wish there was an easy guide for genetic testing?
Now there is with the CMT Genie!
HNF’s CMT Genie is designed to assist you and your health care provider determine what the best option is for you!
MDA Awards Grant to HNF for Groundbreaking Digital Wearable Study on CMT!
Exciting news for the Charcot-Marie-Tooth (CMT) community! The Hereditary Neuropathy Foundation (HNF) has been awarded an Advocacy Collaboration Grant from the Muscular Dystrophy Association (MDA) to further its groundbreaking work in CMT research. This funding will...
Alesta Therapeutics Advances ALE2 to Target Rare Forms of CMT
Early preclinical research indicates that ALE2 could effectively slow or halt the progression of these debilitating symptoms, offering new hope for patients with these rare forms of CMT.
Update on NMD Pharma Clinical Trial
NMD Pharma Initiates Phase 2 Study of NMD670 in Patients with Charcot-MarieTooth Disease Type 1 and 2 NMD Pharma A/S, a clinical-stage biotech company dedicated to developing novel and improved treatments for patients living with neuromuscular diseases, announces that...
HNF’s Wearable Tech Study Delivers Strong Results for CMT Clinical Trials
Stay tuned: Phase 2 Study results to be released late Summer 2025! HNF’s Wearable Tech Study Delivers Strong Results for CMT Clinical Trials The Hereditary Neuropathy Foundation (HNF) is breaking new ground in Charcot-Marie-Tooth (CMT) research with a...
Seeking Participants for a New CMT Clinical Trial: A Chance to Help Advance Treatment
Seeking Participants for a New CMT Clinical Trial (for the US only): A Chance to Help Advance Treatment! The sponsor of the study is NMD Pharma A/S. If you’re living with Charcot-Marie-Tooth (CMT) disease, specifically CMT1 or CMT2 subtypes, you may be eligible to...
Digital Health Technologies for Charcot-Marie-Tooth (CMT) Disease
Digital Health Technologies for Charcot-Marie-Tooth (CMT) DiseasePatients attending the Clinical Trial Readiness Summit will be eligible to participate in this two-week study. All participants will join the clinical researchers and HNF staff for a private dinner on...
Breaking News for SORD Deficiency Patients
The INSPIRE trial is a Phase 3 double-blind placebo-controlled registrational study evaluating the effect of once-daily (QD) oral govorestat (AT-007) in 56 patients aged 16-55 with SORD Deficiency in the US and Europe.
Breaking News: HNF to Host “Revolutionizing Clinical Trials” Panel at Bio 2024
HNF is thrilled to announce that we have been accepted to host a 1-hour panel discussion during the Bio International Convention in San Diego, CA, in June 2024.
Pediatric CMT Natural History Study Enrollment Opening at Two Centers of Excellence
Pediatric CMT Natural History Study enhancement to their Charcot-Marie-Tooth (CMT) and Inherited Neuropathies (IN) patient registry, Global Registry for Inherited Neuropathies (GRIN).
DTx Pharma Receives FDA Orphan Drug Designation for DTx-1252 for the Treatment of Charcot-Marie-Tooth Disease Type 1A (CMT1A)
Dtx today announced that the FDA has granted Orphan Drug Designation to DTx-1252, an investigational FALCON small interfering RNA (siRNA) therapeutic for the treatment of Charcot-Marie-Tooth Disease Type 1A (CMT1A).