HNF![]() recently entered into a partnership with the French biopharmaceutical company, Pharnext, to help raise awareness of Charcot-Marie-Tooth (CMT) disease and support the CMT patient community through several initiatives. Building awareness is key! Pharnext’s support will assist HNF in distributing HNF’s CMT Update quarterly newsletter, enhancing the Global Registry for Inherited Neuropathies (GRIN), setting up activities for CMT September Awareness Month in the US and strengthening the CMT Inspire Community. Pharnext is an advanced clinical stage biopharmaceutical company discovering and developing new therapeutics that target multiple key disease pathways for orphan and common neurological diseases including Charcot-Marie Tooth disease. The company’s lead program PXT-3003 for the treatment of CMT 1A has completed Phase 2 clinical trials and pending discussions with the appropriate regulatory agencies will undergo Phase 3 clinical trials. PXT-3003 was recently granted Orphan Drug designation from the European Medicines Agency and the U.S. Food and Drug Administration. Learn more at www.pharnext.com or www.hnf-cure.org.
recently entered into a partnership with the French biopharmaceutical company, Pharnext, to help raise awareness of Charcot-Marie-Tooth (CMT) disease and support the CMT patient community through several initiatives. Building awareness is key! Pharnext’s support will assist HNF in distributing HNF’s CMT Update quarterly newsletter, enhancing the Global Registry for Inherited Neuropathies (GRIN), setting up activities for CMT September Awareness Month in the US and strengthening the CMT Inspire Community. Pharnext is an advanced clinical stage biopharmaceutical company discovering and developing new therapeutics that target multiple key disease pathways for orphan and common neurological diseases including Charcot-Marie Tooth disease. The company’s lead program PXT-3003 for the treatment of CMT 1A has completed Phase 2 clinical trials and pending discussions with the appropriate regulatory agencies will undergo Phase 3 clinical trials. PXT-3003 was recently granted Orphan Drug designation from the European Medicines Agency and the U.S. Food and Drug Administration. Learn more at www.pharnext.com or www.hnf-cure.org.
Collaboration is the Key to HNF Success
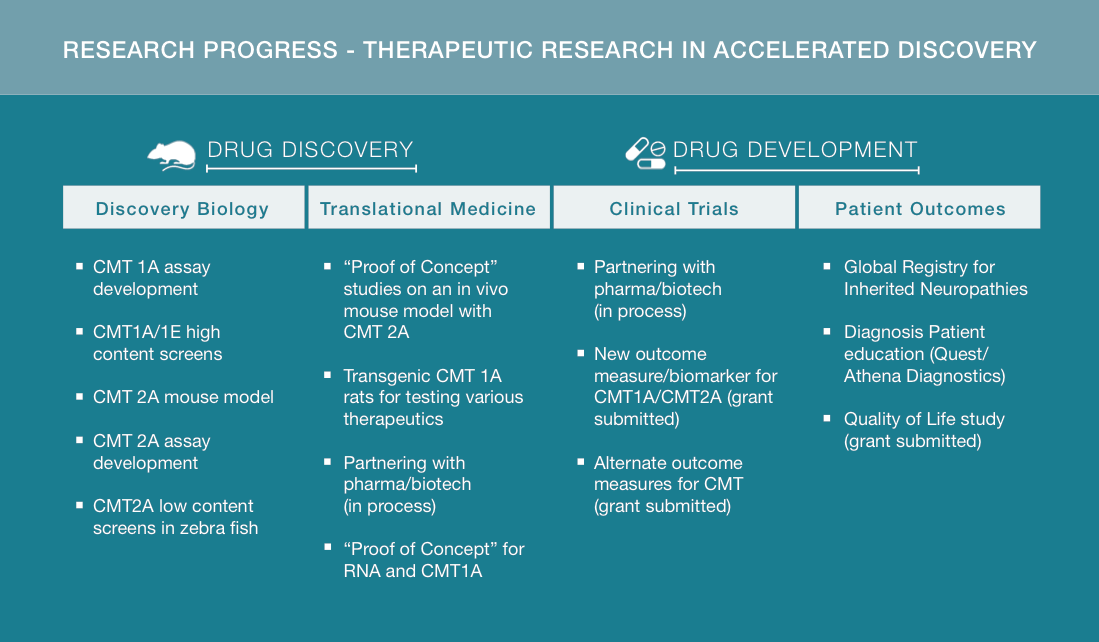
We at HNF are so proud of the accomplishments of those scientists we fund and are asking you to please continue to support our efforts. Our Therapeutic Research In Accelerated Discovery (TRIAD) program is a proven collaborative model in the drug discovery process.




Millions suffer from acute or chronic pain every year and the effects of pain exact a tremendous cost on our country in health care costs, rehabilitation and lost worker productivity, as well as the emotional and financial burden it places on patients and their families. While acute pain is a normal sensation triggered in the nervous system to alert you to possible injury and the need to take care of yourself, chronic pain is different. Chronic pain persists. Pain signals keep firing in the nervous system for weeks, months, even years. Pain affects more Americans than diabetes, heart disease and cancer combined. According to the Institute of Medicine of The National Academies chronic pain affects 100 million Americans. Nyloxin provides all-natural anti-inflammatory analgesic pain relief that is non-toxic, non-narcotic, non-addictive, non-steroidal and contains no aspirin or acetaminophen. Safe to use without a prescription, it treats conditions that cause chronic pain. It’s the only thing that has helped with my CMT and with my Occipital Neuralgia.Help those that are suffering and get paid to do so or use as a means of fundraising for more research!